Introduction
Large granular lymphocytic leukemia (LGLL) is a rare chronic lymphoproliferative disease of T cell and natural killer (NK) cell lineage. Less than 10% of cases are NK-large granular lymphocytic leukemia (NK-LGLL), which is distinguished by clonal growth of mature NK cells that are CD3-, CD16+, and/or CD56+. There are two types of mature NK cells based on CD56 intensity. The CD56dim NK-cell subset exhibits a higher level of natural cytotoxic activity compared to the CD56bright NK-cell subset, which, in turn, is capable of producing abundant cytokines ( Trends Immunol. 2001;22(11):633-640). However, few studies have explored the internal heterogeneity of NK-LGLL based on CD56 expression ( Leukemia. 2010;24(4):881-884; Blood Cancer J. 2018;8(6):51) .
Method
A total of 402 patients have been diagnosed with LGLL at our hospital since June 2005, including 40 with NK-LGLL. All patients met the recommended diagnostic criteria ( Blood. 2017;129(9):1082-1094). Treatment responses were collected after at least 3 months of therapy. NK subtype was defined by flow cytometry analysis using common surface markers showing CD3−/CD16+, or CD56+ pattern, and cells were considered antigen partial positive if antibody staining was between 20%-80%. To perform subgroup analysis, we incorporated partial CD56 positive into the negative group since mature NK cells are CD56-positive. Clonality was evaluated by killer-cell immunoglobulin-like receptors (KIR). Sequencing of STAT3 and STAT5b was performed with Next Generation Sequencing.
Results
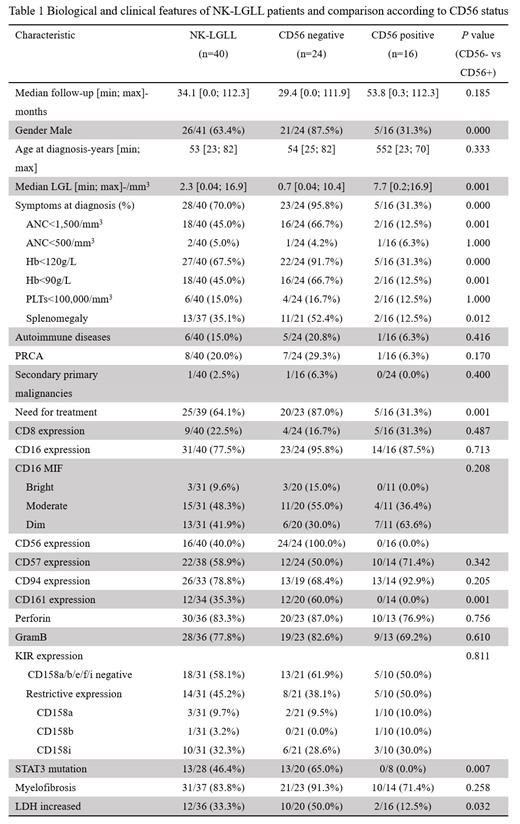
Twenty-eight of 40 patients (70.0%) presented symptoms at the time of diagnosis in which anemia was the most prevalent accounting for 67.5%. PRCA was the most common comorbidity, affecting 20.0% of patients. All cases were CD2+, CD3-. Expressions of mature NK cell markers are heterogenous, including CD8 (22.5%), CD16 (77.5%), CD56 (40.0%), CD57 (58.9%), CD94 (78.8%), CD161 (35.3%), perforin (83.3%), GramB (77.8%). KIR analyses were available in 31 cases, 45.2% of NK-LGLs showed a form of restrictive expression, mainly CD158i (32.3%), an activating KIR, whereas 58.1% of cases lacked CD158a/b/e/f/i expression. STAT3 mutated status was studied in 28 cases (70.0%), 46.4% had STAT3 mutations, and no STAT5B mutations were detected.
We classified 40 cases into two subsets based on CD56 expression (Table 1). CD56- cases displayed a more aggressive course. They usually presented with symptomatic disease including anemia (91.7% vs 31.3%, P= 0.000), severe anemia (66.7% vs 12.5%, P= 0.001), neutropenia (66.7% vs 12.5%, P= 0.001), splenomegaly (52.4% vs 12.5%, P= 0.012), and treatment requirement (87.0% vs 31.3%, P= 0.001). CD56-negative cases expressed CD161 at a higher frequency (60.0% vs 0.0%, P=0.001) while the other mature markers were similar. Interestingly, STAT3 mutation was detected in 13 of 20 (65.0%) in CD56- NK-LGLL, and none of 8 CD56+ NK-LGLL had STAT3 mutation (p=0.007). The treatment pattern was similar between CD56+ vs CD56- NK-LGLL. However, the treatment efficacy was lower in CD56- NK-LGLL, with overall response rates (ORR) of 100% vs 79% and CR (complete response) rate of 80% vs 46%, in CD56+ and CD56- NK-LGLL patients respectively.
Conclusions
NK-LGLL is a heterogenous entity and can be divided into two groups further based on CD56 expression. CD56- NK-LGLL showed more prominent clinical symptoms, high STAT3 mutation, and CD161 expression. CD56- NK-LGLL showed a poor response than CD56+ NK-LGLL. More investigations are deserved to explore the underlying mechanisms.
Disclosures
No relevant conflicts of interest to declare.